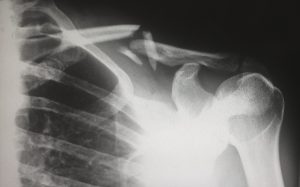
Finnish medical technology developer Bioretec has made history with its RemeOs metal screws, which hasten the healing of broken bones by helping to properly secure them during the often-lengthy mending process. The company says that the inserts are the very first bioresorbable metal implants to ever earn approval from the FDA. The metallic properties of the screws makes them stronger, with added rigidity and hardness, compared to more common polymer-based bioresorbable implants. Moreover, the screws can be leveraged in heavy load-bearing applications, making them a viable option for feet or ankle bone placement.
Instead of consisting of the rare-earth metals that can usually be found in surgical hardware, RemeOs screws are constructed from three elements that are native to the human body—calcium, zinc, and magnesium. Bioretec’s endgame is to end the use of traditional titanium and steel implants by offering an alternative that needn’t be removed after implantation, as its new screws simply shrink to be replaced by new bone.
The company’s research into the orthopedic solutions space showed that the U.S. represents around 65% of the international market. “We are excited that we can now begin to establish key collaborations with hospitals and clinical professionals specialized in ankle fractures in the U.S.,” said Timo Lehtonen, CEO of Bioretec. He also indicated there are plans in place for more implants making use of the RemeOs material down the line.