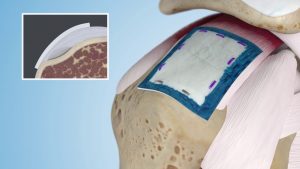
Smith & Nephew’s Regeneten Bioinductive Implant, a postage stamp-sized device designed to disrupt the progression of rotator cuff disease, has received the CE mark of approval. The device has been implanted over 40,000 times in the U.S., and will now cross the ocean to alleviate rotator cuff pain in Europe.
The collagen-based implant is derived from purified bovine achilles tendon, and is designed to cultivate a healing environment. It disrupts disease progression and stimulates the body’s healing response to facilitate new tendon growth. The implant is completely reabsorbed by the body within 6 months.
“We are delighted to gain CE mark for Regeneten and be able to offer up this ground-breaking technology to our customers in Europe,” said Smith & Nephew senior director marketing EMEA Terry Byca. “The U.S. market has demonstrated over the last 5 years that Regeneten is changing surgeons’ traditional approach to rotator cuff repair; biological healing is imperative.”
The implant can be used to repair partial-thickness tears to large (3-5 cm) tears, or even what’s classified as massive tears (those over 5 cm). Only a single small incision over the rotator cuff and the injured tendon is needed to put the device in place. The implant will soon be available to help the 310,000 patients per year who require rotator cuff procedures in Europe, in a market estimated to be worth $175 million annually.
Regeneten hopes to revolutionize a condition that was previously considered ongoing and very costly. Rotator cuff disease can turn into a chronic ailment that puts the patient through a lot of pain. Before being reabsorbed, the highly porous Regeneten implant leaves a layer of newly grown tendon in its place, removing the strain from the rotator cuff.
So far, the device has achieved high clinical efficacy, with new tendon-like tissue growing in all patients, no foreign body or inflammatory reactions, and zero implant-related complications. Patient satisfaction rate is 94% after one year, with an average recovery rate of only 23 days in a sling.