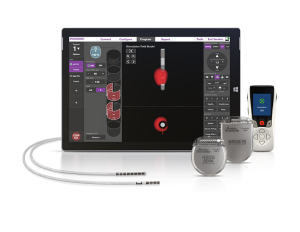
Boston Scientific has honed new software to provide healthcare technicians with a 3D view of the neurostim placement and programming process. With its recent FDA approval, its Stimview XT technology is now poised for a U.S. debut. The agency’s thumbs up comes almost two and a half years after the software earned a CE mark clearance in Europe.
Boston Scientific has placed the software in its existing Vercise Neural Navigator platform, which has been a useful means of assisting the placement and programming of DBS (deep brain stimulation) electrodes. Thanks to Stimview XT, the process will now have invaluable 3D image guidance. As they navigate through the anatomy of an individual’s brain, physicians will leverage the resulting clear, real-time imagery to make sure the lead is correctly oriented and to oversee the placement of electrodes in the correct areas of the brain.
The journal Frontiers in Neurology found that Stimview XT’s image-guided tech “drastically reduces programming time without compromising symptom control and patient satisfaction” when used to commence a DBS regimen in patients with Parkinson’s disease. Ten study subjects received programming through the XT system and typical clinical-based methods, with researchers discovering that physicians could reduce their neurostimulation adjustment time by 56% when compared with the usual technique.